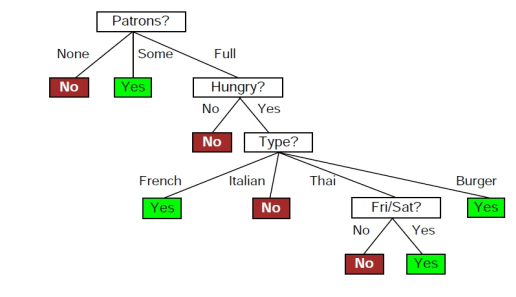
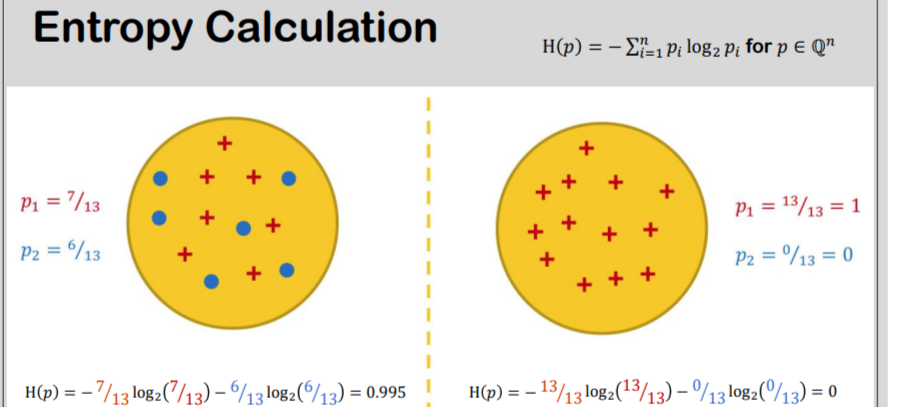
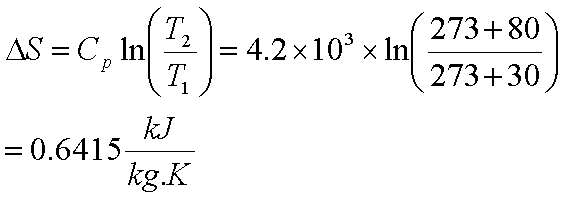information theory - How to calculate conditional entropy using using this tabular probability distribution? - Mathematics Stack Exchange

Calculation of Configurational Entropy with a Boltzmann–Quasiharmonic Model: The Origin of High-Affinity Protein–Ligand Binding | The Journal of Physical Chemistry B

SOLVED: Calculate the entropy change of sublimation of ice, H2O(s) to H2O(g) at the temperature of the triple point of water, 273.16 K. Use the necessary data from the Resource Section.

Calculate the entropy change of n-hexae when 1mol of it evaporates at 341.7K (Delta(vap)H^(Theta) = 290.0 kJ mol^(-1))

Calculate the entropy change for the following reaction, H2(g) + Cl2(g)⟶ 2HCl(g) at 298 K . Given that, S^ H2 = 131 J K^-1 mol^-1 , S^ Cl2 = 223 J K^-1 mol^-1 and S^ HCl = 187 J K^-1 mol^-1 .




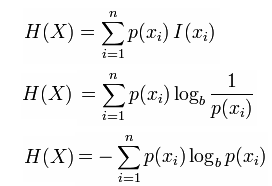






![15.2 Calculate the standard entropy change for a reaction [HL IB Chemistry] - YouTube 15.2 Calculate the standard entropy change for a reaction [HL IB Chemistry] - YouTube](https://i.ytimg.com/vi/IwRy4iYVQLI/maxresdefault.jpg)